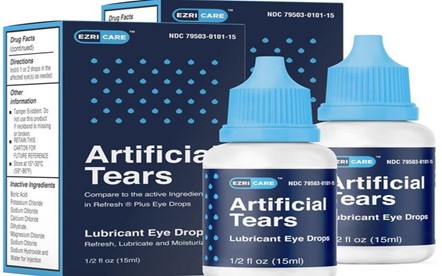
The National Agency for Food and Drugs Administration and Control (NAFDAC) has notified the public about the severe adverse effects of EzriCare and Delsam Pharma Artificial Tears Lubricant Eye Drops.
NAFDAC’s Public Alert No:008/2023 noted that the eye drops were linked to death and blindness in the United States.
Global Pharma Healthcare, a pharmaceutical company situated in Tamilnadu, India, manufactures the harmful eye drop products.
READ ALSO: Japata Bitters, a Nigerian Alcoholic Drink, Kills Mice at Testing
“The National Agency for Food and Drugs Administration and Control (NAFDAC) is notifying the public that Global Pharma Healthcare recalled EzriCare and Delsam Pharma Artificial Tears eye drops early in February. The recall of the preservative-free eye drops were due to suspected contamination with pseudomonas aeruginosa, a highly resistant bacteria,” NAFDAC stated on Wednesday.
“Since the recall, the US Centre for Disease Control (CDC) has reported 68 patients across 16 states who have suffered health problems from the bacteria, including eye infections and permanent loss of vision. Surgical removal of eyeballs and a case of death with a ‘bloodstream infection’ have also been reported.
“Laboratory testing identified the bacteria in opened EzriCare bottles from multiple sources, according to the CDC. Unopened bottles are currently being examined to check whether contamination occurred during manufacturing.
“Pseudomonas is a type of bacteria (germ) that is found commonly in the environment, like in soil and in water. Of the many different types of Pseudomonas, the one that most often causes infections in humans is called Pseudomonas aeruginosa, which can cause infections in the blood, lungs (pneumonia), or other parts of the body after surgery.
“Signs of infection include yellow, green, or clear discharge from the eye, discomfort or pain, redness, blurry vision and increased sensitivity to light.
“Artificial Tears (carboxymethylcellulose sodium) Lubricant Eye Drops are used as a protectant against further irritation or to relieve dryness of the eye for the temporary relief of discomfort due to minor irritations of the eye, or exposure to wind or sun.”
NAFDAC has asked the public not to use the eye drops.
READ ALSO: Ambronol and DoK-1 Max, two Syrups Manufactured in India, Can Kill
NAFDAC also said that the product was not on its database and advised importers, distributors, retailers and consumers to avoid buying or selling the eye drops.
Anyone in possession of the eye drops should submit their stock to the nearest NAFDAC facility.
Subscribe
Be the first to receive special investigative reports and features in your inbox.